Medical imaging is used for eligibility, safety and efficacy endpoints in clinical trials. The Bracken team builds on years of strategic and operational experience to help clients develop and design the imaging aspects of their clinical trials.
We offer comprehensive support in biomarker selection and development, CRO/vendor selection, biopharmaceutical products, novel diagnostic imaging products, precision medicine, managing core labs and vendors, patient-focused drug development, and clinical and imaging strategy and protocol development.
Bracken’s integrated approach to medical imaging offers support that’s not just intellectual, it’s interpersonal.
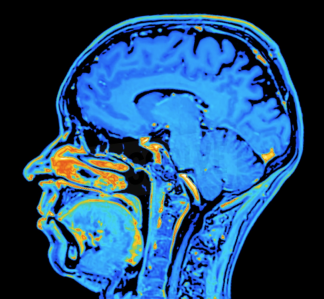
Endpoints play a pivotal role in advancing precision medicine, but their true value depends on the quality of medical image acquisitions and the expertise of radiologists interpreting them.
The experience of working at the intersection of medical imaging and clinical research has provided the Bracken consulting team with the knowledge and insight to help you:
Bracken can assist you in identifying the imaging core labs that best meet your requirements.
How? By asking the right questions:
Most core labs lack staff with direct experience in the pharma and biotech industries, limiting their nuanced understanding of critical decision-making.
Similarly, most pharma teams have limited expertise in medical imaging.
Bracken is here to bridge these gaps in knowledge.
In this webinar, guest speaker and Bracken CEO Dr. Colin Miller shows how early-stage data can shape smarter trial designs and help you stay ahead of evolving imaging demands, enhance study outcomes, and meet regulatory expectations with confidence.
We have deep experience with designing and planning clinical trials that succeed from both a scientific and a commercial perspective.
We would be delighted to hear from you and discuss our consulting services. Please call +1 215 648 1208 to chat with us directly. Or write to us using the form provided here. We look forward to collaborating with you.
Subscribe to our monthly hand-crafted newsletter for the latest insights on clinical trial trends and the life science industry.
12 Penns Trail, Newtown, PA 18940
+1 215 648 1208
© 2026 the bracken group - Privacy Policy
12 Penns Trail, Newtown, PA 18940
+1 215 648 1208
© 2026 the bracken group - Privacy Policy